News Archive
Updated List of IVDR-Certified Products
We have successfully obtained our certification under the new In Vitro Diagnostic Regulation (IVDR; 2017/746)! The first batch of flagship products are already available on the market under the new IVDR, well in advance of the transition period deadline. Read more.
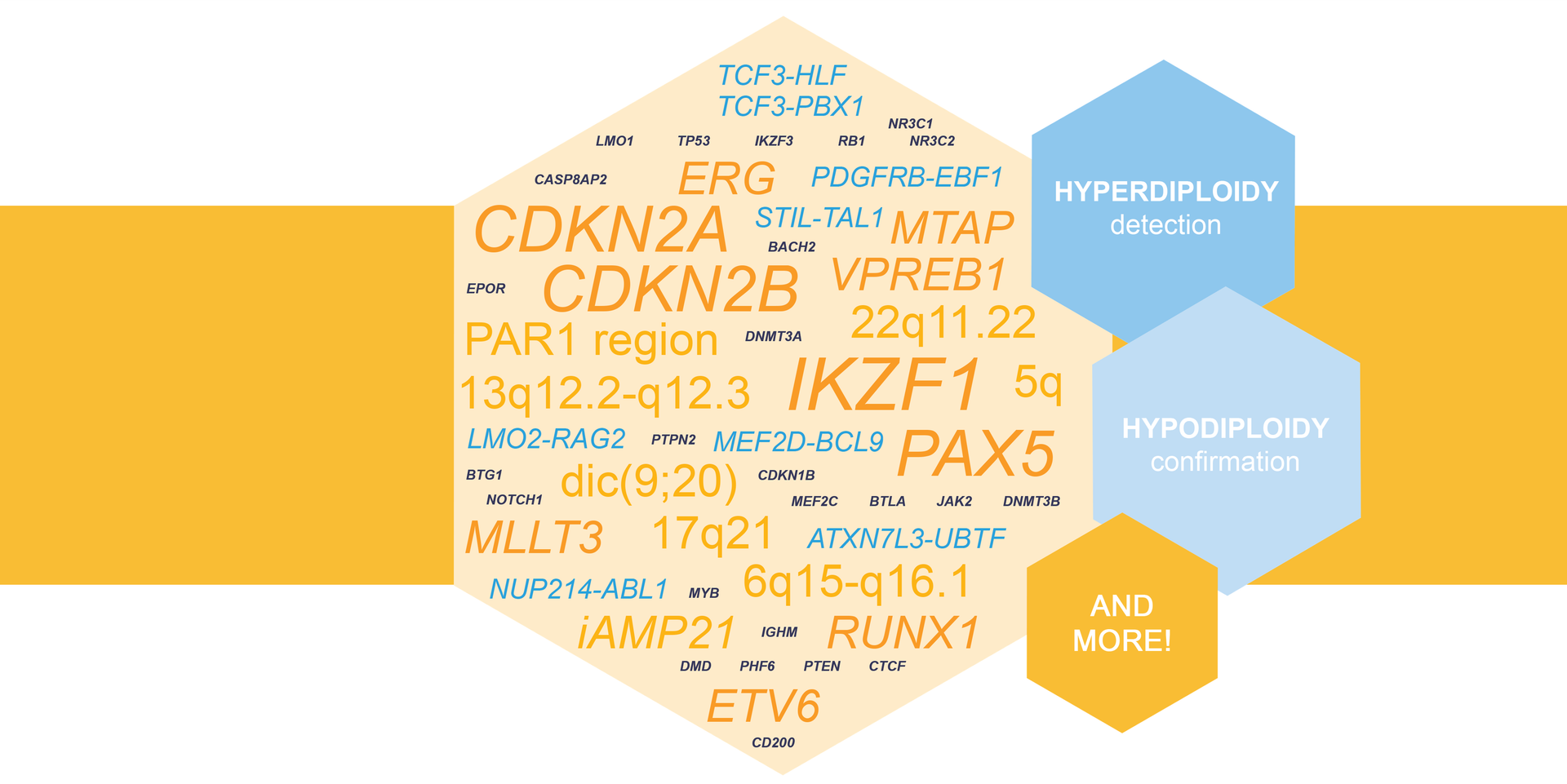
digitalMLPA as a Valuable Tool in Adult ALL Risk Stratification
D007 Acute Lymphoblastic Leukemia was recognized as a valuable tool for enhanced molecular testing of adult ALL cases in work shown recently at a scientific congress. Read more.
Test Labs Wanted: digitalMLPA NXtec D011 Cardiac Panel
The external test phase for digitalMLPA assay NXtec D011 Cardiac Panel is about to start, and we are looking for test labs. Read more.
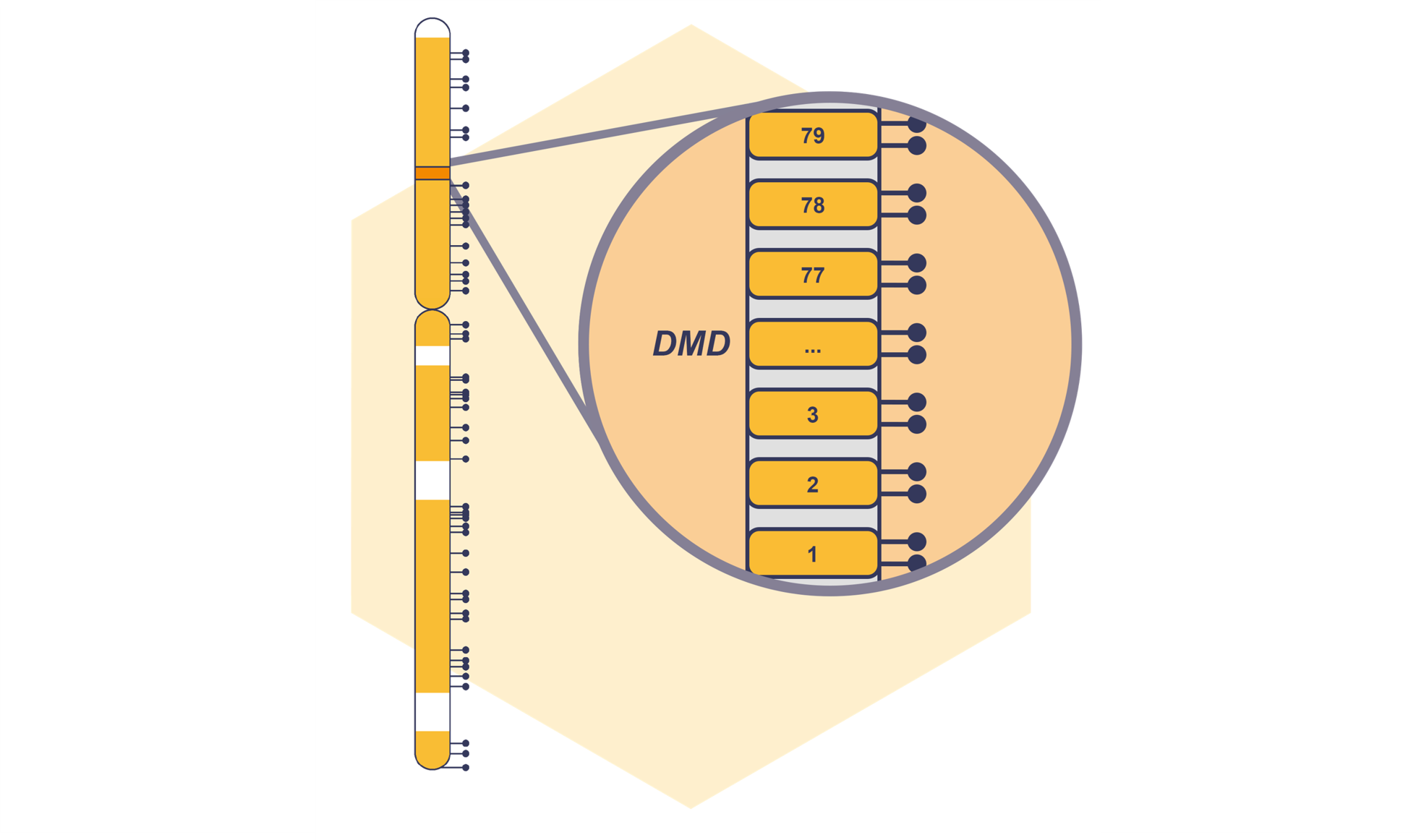
Coming Soon: NXtec D008 DMD for Comprehensive CNV Detection in DMD
Our digitalMLPA™ portfolio is about to receive an update with the upcoming launch of NXtec D008 DMD – our first dedicated neuromuscular digitalMLPA assay. Read more.
Let's Connect at ESHG 2026
Visit us at booth 404 during ESHG 2026, taking place 13-16 June in Gothenburg (Sweden), and discover how our products can support critical clinical questions and accelerate your research. Read more.
digitalMLPA Uncovers CNVs in Hereditary Cancer Cases Missed by NGS Alone
A recent study by researchers at the Hacettepe University highlights how D001 Hereditary Cancer Panel 1 provide clinically relevant insights where standard NGS falls short. Read more.
Wanted: DNA Samples
MRC Holland is looking for DNA samples for the development and quality control of our many products.
Updates to our DMD Portfolio
We are pleased to share an important update to our Duchenne muscular dystrophy (DMD) and Becker muscular dystrophy (BMD) portfolio. Improved versions of our MLPA assays for DMD copy number analysis are now available, offering enhanced performance and quality control. Read more.
New Product: NXtec D028 Carrier Panel 1
We are proud to introduce NXtec D028 Carrier Panel 1 - a high-throughput assay detecting CNVs and selected SNVs for carrier status analysis. Read more.
A Year in Review – Reflecting on 2025
As we look back on 2025, we are excited to share some of the year’s highlights with you! Read more.