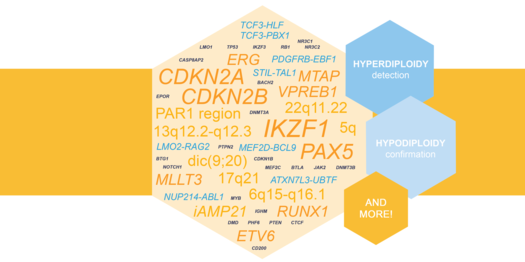
Highlighted

Let's Connect at ESHG 2026
Visit us at booth 404 during ESHG 2026, taking place 13-16 June in Gothenburg (Sweden), and discover how our products can support critical clinical questions and accelerate your research. Read more.
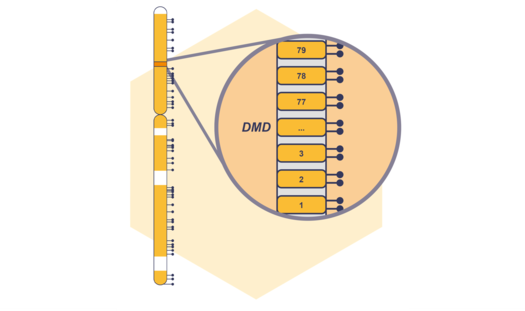
Coming Soon: NXtec D008 DMD for Comprehensive CNV Detection in DMD
Our digitalMLPA™ portfolio is about to receive an update with the upcoming launch of NXtec D008 DMD – our first dedicated neuromuscular digitalMLPA assay. Read more.
Test Labs Wanted: digitalMLPA NXtec D011 Cardiac Panel
The external test phase for digitalMLPA assay NXtec D011 Cardiac Panel is about to start, and we are looking for test labs. Read more.