Newborn Screening: Spinal Muscular Atrophy (SMA)
Why screen newborns for Spinal Muscular Atrophy (SMA)?
 SMA is a life-threatening genetic neuromuscular disorder that affects the nerve cells controlling the muscles. SMA results in neuronal degeneration and muscular atrophy. Worldwide, SMA affects 1 in 6,000–10,000 live births. There are four types of SMA, distinguished by age of disease onset and severity. The most severe type, type 1, has an onset before 6 months of age and those affected, if left untreated, typically do not survive past two years of age.
SMA is a life-threatening genetic neuromuscular disorder that affects the nerve cells controlling the muscles. SMA results in neuronal degeneration and muscular atrophy. Worldwide, SMA affects 1 in 6,000–10,000 live births. There are four types of SMA, distinguished by age of disease onset and severity. The most severe type, type 1, has an onset before 6 months of age and those affected, if left untreated, typically do not survive past two years of age.
The increased availability of SMA treatments has led to a corresponding interest in the addition of SMA to national newborn screening programs. This is especially important as the best outcomes for SMA are achieved when treatment is started presymptomatically.
What causes SMA?
SMA is caused by having an insufficient amount of survival motor neuron (SMN) protein in cells. The SMN protein ensures that motor neurons, responsible for transferring signals from brain to muscle cells, remain healthy. If the SMN protein is lacking or present at a very low level, motor neurons die and muscles wither away.
The SMN protein is primarily produced from the SMN1 gene. SMA patients have no functional copies of the SMN1 gene from which SMN protein can be produced. People with one SMN1 copy are SMA carriers: they do not have SMA symptoms. However, a carrier couple may have children with SMA.
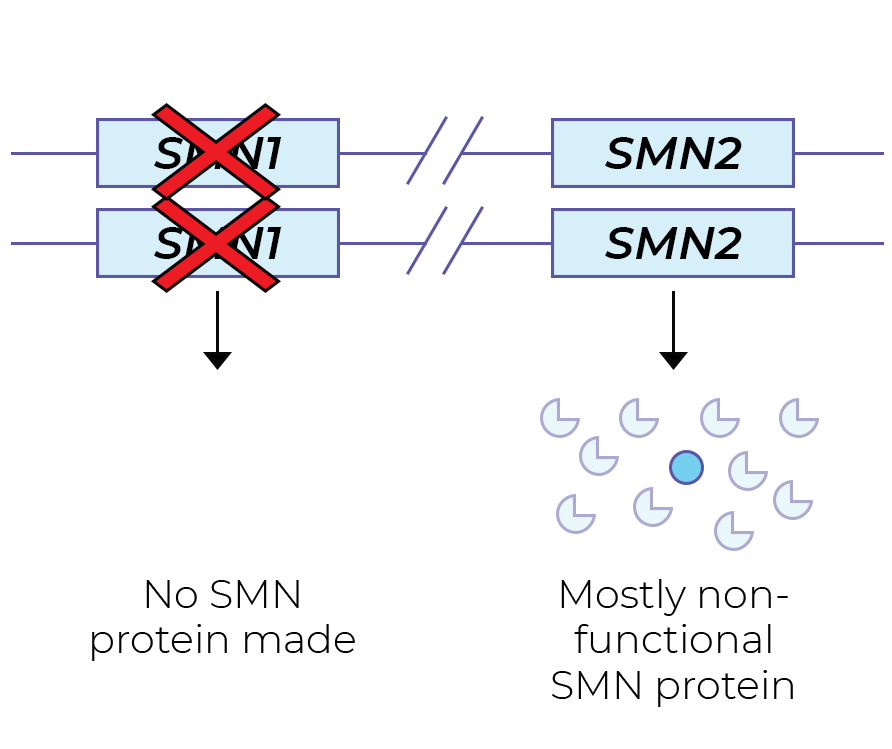
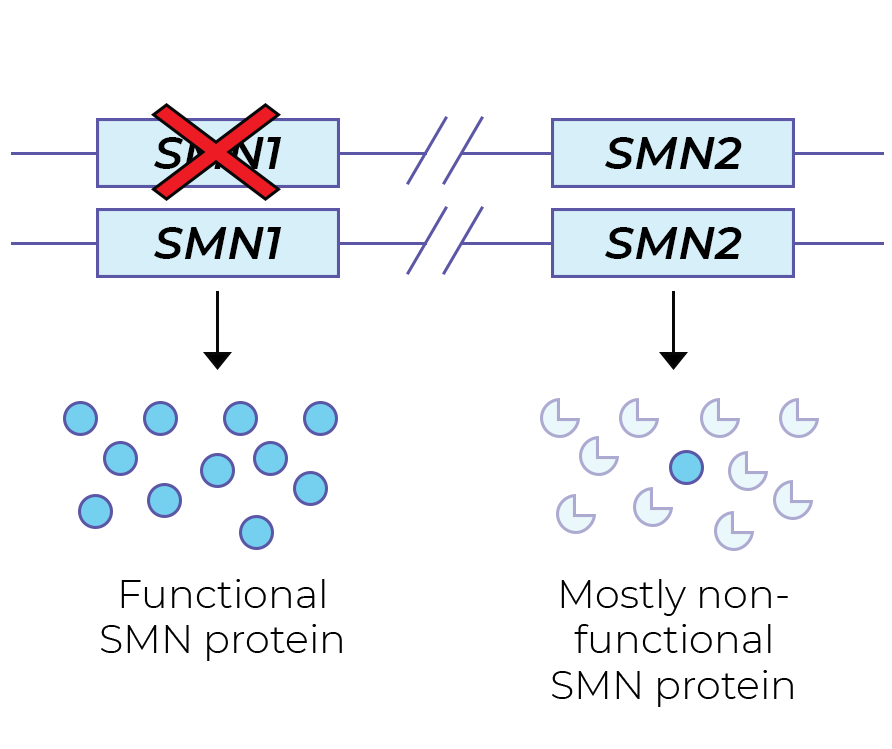
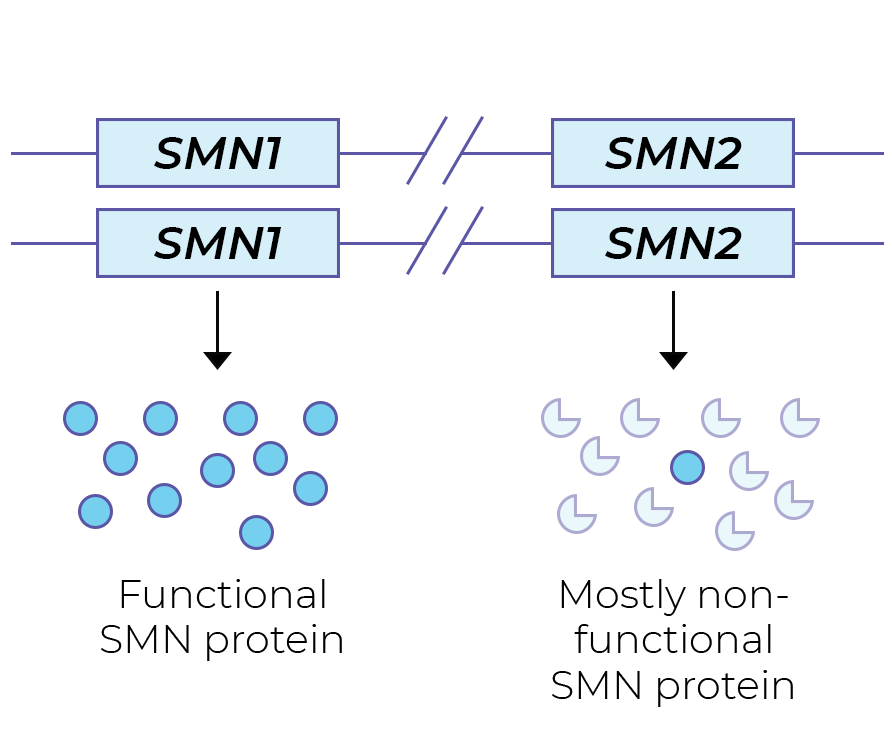
The highly similar SMN2 gene also plays a role in SMA. SMN2 mostly generates non-functional SMN protein, and a tiny amount of functional SMN protein. This difference in SMN1 and SMN2 protein stability is the result of a single nucleotide difference (a C>T change in exon 7), resulting in different splicing.
SMN2 copy numbers vary between individuals in the population. For SMA patients, the more SMN2 copies they have, the less severe their symptoms tend to be. Quantifying SMN2 copies is therefore important for disease prognosis. In addition, a patient's SMN2 copy numbers may influence their eligibility for treatment.
SALSA® MC002 SMA Newborn Screen: MRC Holland's solution for SMA neonatal screening
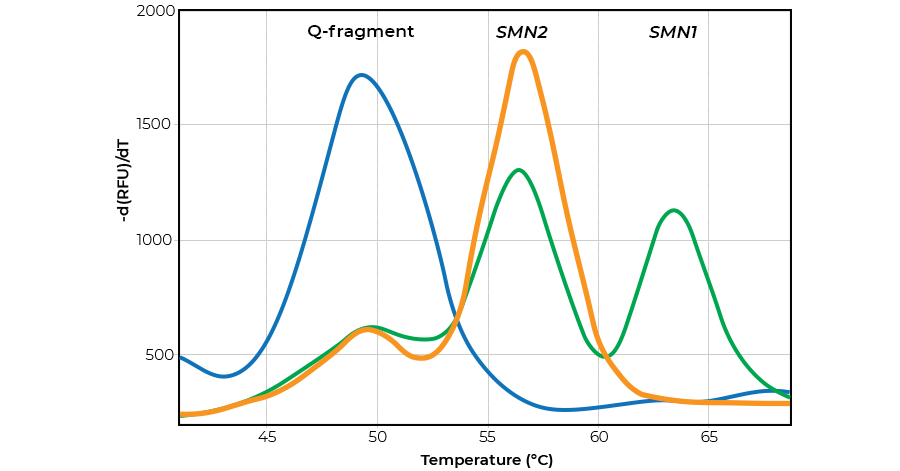 MRC Holland's SALSA MC002 SMA Newborn Screen is based on melt curve analysis: a simple and affordable technique that utilises the fact that different genetic sequences have different DNA melting temperatures. Using MC002's amplification and probe binding-based approach, peaks specific for SMN1 and SMN2 are generated. The method is highly sequence specific, sensitive, and easy to perform. MC002 accurately determines the presence or absence of the SMN1 and SMN2 gene, reliably identifying SMA patients (0 SMN1 copies) but not symptom-free carriers (1 copy).
MRC Holland's SALSA MC002 SMA Newborn Screen is based on melt curve analysis: a simple and affordable technique that utilises the fact that different genetic sequences have different DNA melting temperatures. Using MC002's amplification and probe binding-based approach, peaks specific for SMN1 and SMN2 are generated. The method is highly sequence specific, sensitive, and easy to perform. MC002 accurately determines the presence or absence of the SMN1 and SMN2 gene, reliably identifying SMA patients (0 SMN1 copies) but not symptom-free carriers (1 copy).
Confirmation of MC002 results on patients' dried blood spot (DBS) extracts can be done using the MLPA assay P021 SMA. With P021 SMA, SMN2 copy number is also directly determined for quick prognosis and treatment.
MRC Holland's MLPA assays are the worldwide market leader in diagnostic tests for SMA. MRC Holland's latest arrival, SALSA MC002 SMA Newborn Screen, was developed in close collaboration with top institutes involved in neonatal screening and SMA diagnosis.
Comparison of SMA products available from MRC Holland
| MC002 | P021 | P060 | P460 | D028 | ||
|---|---|---|---|---|---|---|
| Properties | CE-marked | yes | yes | yes 1 | yes | no |
| Technique | Melt Assay | MLPA | MLPA | MLPA | digitalMLPA | |
| Used for | Neonatal Screening | ● | ○ | |||
| Patient Detection | ● | ○ | ○ | ○ | ||
| Carrier Detection | ○ | ● | ● | ● | ||
| Silent Carrier 2 Detection | ● | ● | ||||
| Coverage | SMN1 exon 7 | ✓ 3 | ✓ | ✓ | ✓ | ✓ |
| SMN1 exon 8 | ✓ | ✓ | ✓ | ✓ | ||
| SMN2 exon 7 | ✓ 3 | ✓ | ✓ 1 | ✓ | ||
| SMN2 exon 8 | ✓ | ✓ 1 | ||||
| SMN1+2 exon 1–8 | ✓ | |||||
| Silent Carrier SNP Probe(s) | ✓ | ✓ | ||||
| Sample type for diagnostic purposes |
Peripheral whole blood | ✓ | ✓ | ✓ | ✓ | |
| Dry blood spot (DBS) cards | ✓ 4 | ✓ 4 | ||||
| Product page | MC002 | P021 | P060 | P460 | D028 | |
● Primary application.
○ Secondary application.
1 Probes for SMN2 in P060 are intended to be used as interpretation aid for SMN1 copy number determination. For SMN2 copy number determination we recommend using P021 SMA.
2 MC002: no absolute copy numbers aside from 0 determined.
3 Increased detection of silent carriers: individuals with 2 SMN1 copies on one allele + 0 on the other.
4 MC002 SMA Newborn Screen: crude extract from washed or unwashed DBS cards; P021 SMA: crude extract from washed DBS cards only.
More Information
Read our flyer about MC002.